Virtually no use for the pure metal, however two of its compounds lime (CaO) and gypsum (CaSO4) are in great demand by a number of industries. 
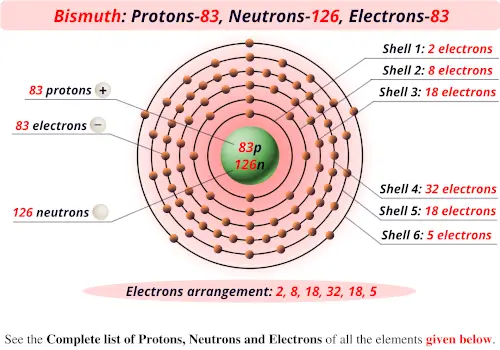
6 of them are naturally occurring, 5 of which are stable.Ĭalcium is used by many forms of life to make shells and bones. Thus, the calculations in this section are applied as a general approximation for elements from helium (2 protons) to calcium (20 protons), but they are using. Calcium is an essential element for living organisms. Protons Electrons Charge Name Ca 20 20 0 Calcium Ca2+ 20 18 2+ Calcium. 41 Ca has received much attention in stellar studies because it decays to 41 K, a critical indicator of solar system anomalies. 
It is used a reducing agent in the extraction of thorium, zirconium and uranium. CHAPTER 3 COMPOUNDS Ions have an unequal number of protons and electrons. Most of its production is in the upper metre of the soil column, where the cosmogenic neutron flux is still sufficiently strong. This simulation creates an atom from protons, neutrons, and electrons and test skills with the periodic table.įor more information about atoms, please see UC Davis's chem wiki.Calcium is a soft grey metallic element belonging to group 2 of the periodic table. Our team has created a video in the Knowing Nuclear stream about this:īelow is a interactive PhET simulation from the University of Colorado. This is discussed more on the page about the nucleus. For example carbon-12 ( 12C) is one isotope of carbon and carbon-14 ( 14C) is another isotope of carbon. These are referred to as different isotopes of an atom. The same type of atom (determined by number of protons) can have different numbers of neutrons. Subtracting 55 from 133 gives 78, which is thus the number of neutrons within the atom. For example, cesium is number 55 on the periodic table of elements and thus has 55 protons furthermore, its atomic mass (typically also found on the periodic table) is known to be 133 amu (atomic mass units). Simply subtracting the number of protons from the atomic mass of the atom will give the number of neutrons. Neutrons have the same mass as protons, making it very easy to determine how many are within a nucleus of an atom. This radiation causes ions to form and as a result is called ionizing radiation. One way that atoms gain or lose electrons is with high energy radiation. Ions can bond readily to other ions, creating a wide variety of different compounds. If the opposite happens and the atom loses an electron it becomes a positively charged ion or a cation. For example, a carbon atom has exactly 6 protons in its nucleus and is thus number 6 on the periodic table of elements, thorium has exactly 90 protons and is thus number 90 on the periodic table of elements.Ītoms have an equal number of protons and electrons however, an atom can lose or gain electron(s) becoming "unbalanced." An unbalanced atom is called an ion if it gains an electron (thus having more electrons than protons) it becomes a negatively charged ion or an anion. Īn element is identifiable by the number of protons found within a nucleus of one of its atoms (see figure 3) furthermore, the number of protons in the atom also determines the element's location on the periodic table of elements. This is an atom of helium - notice how it has two protons. Note the diameters in the picture give the scale. Atoms have protons and neutrons in their nuclei with electrons orbiting outside. Please check out the scale of the universe to see a visual representation of just how small atoms are.įigure 3. of Protons, Neutrons, Electrons and Key Drawing Bohr Models of Atoms 1 20. In fact, one cubic centimeter of the silicon seen in figure 1 contains approximately 5 x 10 22 atoms (that's 5 with 22 zeros after it!). The number of protons in one atom of an element determines the atomsand the. Differing quantities of protons, neutrons, and electrons cause the atom to have differing chemical properties, which determine what element that atom is.Ītoms are unimaginably small, and their nuclei are 1000 times smaller. 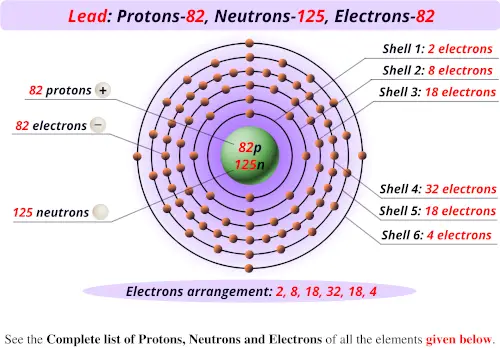
Surrounding the nucleus is a cloud of much smaller and lighter electrons, which are attracted to the nucleus by the electromagnetic force from interacting with the protons. Protons and neutrons are held together in the nucleus by what's called the strong nuclear force (which is the strongest known force in the universe). It is composed of protons and neutrons (collectively called nucleons) and is responsible for the large majority of the atomic mass. The nucleus is the central, highly dense component of an atom (see figure 2). Notice how much more area the electron cloud occupies compared to the nucleus (although this is still not quite to scale, it is close). A drawing depicting what an atom looks like. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
